Executive Summary
Saline industrial wastewater, often containing 3 to 15 % sodium chloride along with organic pollutants such as phenols, hydrocarbons, and aromatic compounds, poses significant challenges to conventional biological treatment systems. Non-halophilic microbes suffer osmotic stress in these environments, leading to plasmolysis and reduced metabolic activity, which results in poor chemical oxygen demand (COD) removal and system instability. Halophilic bacteria and archaea, affectionately known as "salty bugs," however, thrive in these conditions through specialized physiological adaptations.
This article examines the biology and physiology of these organisms, details their salt tolerance and organic degradation mechanisms, and explores their incorporation into integrated fixed-film activated sludge (IFAS) and biofilm systems. Applications demonstrate high removal efficiencies, often exceeding 90 % for key contaminants under continuous operation. Yet full-scale implementation faces hurdles including biomass washout and equipment corrosion. Strategic engineering solutions directly address these physical challenges; for instance, specialized carrier media selection prevents biomass loss, while the use of corrosion-resistant materials protects facility infrastructure. It is my hope you will gain actionable insights for designing robust treatment trains that balance microbial ecology with engineering practicality to effectively clean these complex waste streams.
Table of Contents
- Introduction
- Biology and Physiology of Halophilic Bacteria
- Mechanisms of Salt Tolerance and Organic Degradation
- Applications in IFAS and Biofilm Systems
- Typical Treatment Train Configuration
- Challenges in Full-Scale Implementation
- Engineering Solutions and Best Practices
- Conclusion
- Glossary
- Frequently Asked Questions
- Bibliography
Introduction
Industrial processes generate vast volumes of saline wastewater, particularly from petroleum refining, sauce manufacturing, pharmaceuticals, and seafood processing. These streams typically exhibit total dissolved solids (TDS) levels of 10 to 100 g/L NaCl, accompanied by recalcitrant organics that inhibit standard activated sludge processes. Conventional microbes experience cell dehydration and enzyme denaturation under high ionic strength, often necessitating energy-intensive pretreatment like dilution or evaporation. Halophilic bacteria provide a biological solution by maintaining metabolic activity across wide salinity ranges. Their use dates to laboratory demonstrations in the 1990s, yet full-scale adoption remains limited due to knowledge gaps in reactor design and operational control. This review synthesizes current understanding to equip you with both the conceptual foundations and practical engineering considerations needed to effectively treat and clean these streams.
Biology and Physiology of Halophilic Bacteria
Halophilic microorganisms are classified by their optimal salinity requirements: slight halophiles (1 to 3 % NaCl), moderate halophiles (3 to 15 % NaCl), and extreme halophiles (15 to 30 % NaCl). Bacteria such as Halomonas species dominate moderate groups, while archaea like Haloferax and Halobacterium exemplify extremes. These organisms inhabit natural hypersaline environments including solar salterns, salt lakes, and contaminated industrial brines. Physiologically, they exhibit robust cell walls enriched with negatively charged surface proteins that resist high ionic strength. Growth optima often align with 5 to 10 % NaCl for many industrial strains, with doubling times comparable to mesophilic counterparts under ideal conditions. Many form biofilms or produce extracellular polymeric substances (EPS) that enhance community resilience. Diversity surveys using 16S rRNA sequencing reveal Proteobacteria and Bacteroidetes as prevalent phyla in saline treatment systems.
Mechanisms of Salt Tolerance and Organic Degradation
Halophilic microbes employ two primary strategies for salt tolerance. Extreme halophiles often use the “salt-in” approach, accumulating intracellular potassium chloride (KCl) to balance external osmotic pressure. Their enzymes feature increased acidic amino acids (glutamate, aspartate) for solubility in high salt, rendering them inactive below 10 % NaCl. Moderate halophiles and halotolerant bacteria predominantly adopt the “salt-out” strategy, synthesizing or importing compatible solutes such as ectoine (C6H10N2O2), glycine betaine, trehalose, or hydroxyectoine. These zwitterionic or neutral molecules maintain cytoplasmic water activity without disrupting protein function. Osmotic pressure across the membrane follows the van't Hoff equation: π = iCRT where π is osmotic pressure (bar), i is the van't Hoff factor (≈2 for NaCl), C is molar concentration, R is the gas constant (0.08314 L·bar·mol-1·K-1), and T is absolute temperature (K). At 5 % NaCl (≈0.85 M), π approximates 42 bar at 25 °C, a force that non-halophilic microbes cannot counter without solute accumulation.

Organic degradation relies on adapted catabolic pathways. For aromatic compounds like phenol, aerobic metabolism proceeds via the β-ketoadipate pathway. Phenol is hydroxylated to catechol, then cleaved by catechol 1,2-dioxygenase (ortho pathway) to cis,cis-muconate, ultimately yielding succinyl-CoA and acetyl-CoA that enter the tricarboxylic acid (TCA) cycle. Alternative meta-cleavage or gentisate pathways operate in certain Marinobacter and Haloferax strains. Halophilic enzymes retain activity at high salt because compatible solutes stabilize tertiary structures. Anaerobic routes via benzyl-CoA are less common but documented in some consortia. These mechanisms enable efficient COD removal even at 10 to 20 g/L TDS.
Applications in IFAS and Biofilm Systems for High-Salinity Wastewater
IFAS and biofilm systems excel with halophiles because attached growth provides physical protection via EPS matrices and higher biomass retention than suspended cultures. In IFAS, plastic carriers or fixed media support slow-growing halophilic populations while suspended flocs handle readily degradable organics. Studies report 90 to 99 % phenol removal in continuous biofilm reactors at 5 to 15 % salinity and hydraulic retention times (HRT) of 4 to 10 hours. For example, immobilized Oceanomonas sp. achieved near-complete phenol elimination in a baffled contactor. Aerobic granular sludge augmented with halophiles maintains structural integrity better than conventional activated sludge at >40 g/L NaCl, producing higher alginate-like EPS that enhances settling. Biofilm systems also resist salinity fluctuations better, as diffusion gradients within the matrix buffer osmotic stress. These configurations suit variable industrial flows and enable simultaneous nitrification-denitrification in saline matrices.
Table 1. Performance Comparison of Conventional Activated Sludge versus Halophilic IFAS/Biofilm Systems in High-Salinity Wastewater Treatment
| Parameter | Conventional Activated Sludge | Halophilic IFAS/Biofilm | Key Advantage of Halophilic Approach |
|---|---|---|---|
| Biomass Retention (g VSS/L) | 2 to 4 | 8 to 15 | Reduced washout under shock load |
| COD Removal Efficiency (%) | 40 to 70 (at >5% NaCl) | 85 to 99 | Sustained metabolism at high TDS |
| Salt Tolerance Limit (% NaCl) | <3 | 3 to 25 | No dilution required |
| Settling Velocity (m/h) | 0.5 to 2 | 5 to 15 | Superior sludge characteristics |
| Typical HRT (h) | 12 to 24 | 4 to 10 | Compact reactor footprint |

Typical Treatment Train Configuration
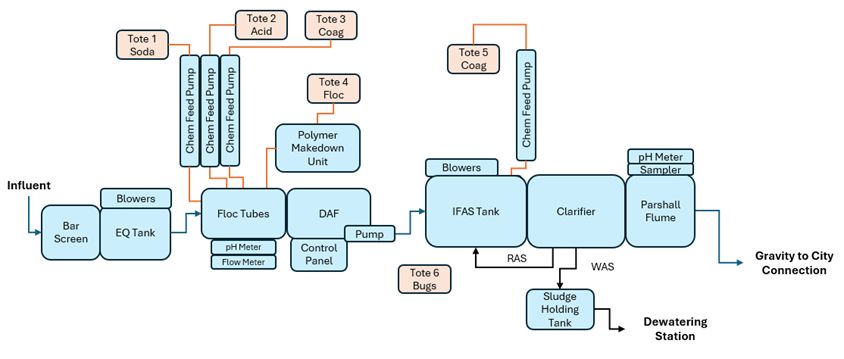
To successfully harness halophilic bacteria in a full-scale setting, the biological reactor must be protected from physical debris and extreme shock loads. A typical treatment train, if there even is one, begins with screening to remove large solids, followed by an equalization (EQ) tank to smooth out flow rates and sudden salinity spikes. Next, the wastewater enters a chemical reaction tank (CRT) for coagulation and flocculation, preparing it for the dissolved air flotation (DAF) unit. The DAF strips out oils, suspended solids, and emulsified contaminants. Only after this rigorous physico-chemical pretreatment does the wastewater enter the halophilic IFAS tank, where our "salty bugs" can efficiently degrade the dissolved organics without being overwhelmed by physical fouling. Finally, a clarifier separates the biological solids from the clean treated effluent.
Challenges in Full-Scale Implementation
Despite promise, scaling halophilic systems encounters several obstacles. Sudden salinity spikes can lyse cells if compatible solute pools are insufficient. Reduced microbial diversity in hypersaline environments may impair nutrient cycling, particularly nitrification, because ammonia-oxidizing bacteria are highly salt-sensitive. Equipment corrosion accelerates due to chloride ions, increasing capital costs for stainless steel or coated materials. Poor floc formation and high EPS can cause foaming or membrane fouling in hybrid systems. Operational expenditures rise if external carbon or compatible solutes are needed for startup. Finally, regulatory acceptance lags because effluent microbial safety and genetic stability require rigorous monitoring.
Engineering Solutions and Best Practices
We address these issues through targeted strategies. Gradual acclimation over 2 to 4 weeks builds robust consortia, while bioaugmentation with pre-adapted Halomonas or Haloferax cultures shortens startup. IFAS media selection favors high-surface-area carriers that promote thick biofilms resistant to shear. Dissolved oxygen control at 1 to 3 mg/L optimizes aerobic degradation without excessive energy use. Process monitoring employs online sensors for conductivity, pH, and volatile fatty acids to enable real-time adjustments. Addition of cheap compatible solutes like betaine precursors during transients enhances resilience. Hybrid configurations, such as a sequencing batch reactor (SBR) with membrane separation, combine biological treatment with polishing. Regular sludge age management (15 to 30 days) maintains active biomass. These practices, validated in pilot trials, reduce overall costs by 20 to 40 % compared with physico-chemical alternatives.

Conclusion
Halophilic bacteria are more than just survivors in harsh environments. They represent a highly specialized biological workforce capable of effectively treating and cleaning challenging saline industrial wastewater. By harnessing their unique mechanisms for salt tolerance and pollutant catabolism and giving them a physical stronghold within IFAS and biofilm architectures, we can achieve reliable treatment performance even under extreme conditions. While real-world hurdles like equipment corrosion and biomass washout persist, engineering innovations rooted in microbial ecology provide clear pathways to full-scale success. Ultimately, the strategic use of these salty bugs ensures that high-salinity treatment plants remain robust, practical, and capable of handling the toughest industrial streams.

Glossary
- Archaea: A domain of single-celled microorganisms distinct from bacteria; many extreme halophiles belong to this group, including Haloferax and Halobacterium.
- Bacteroidetes: One of the two major bacterial phyla, containing a wide range of bacteria that are often found in saline wastewater treatment systems, often contributing to organic degradation.
- Betaine: A compatible solute (glycine betaine) that helps halophilic and halotolerant bacteria maintain osmotic balance without interfering with cellular metabolism.
- Biofilm: A structured community of microorganisms attached to a surface and embedded in a self-produced EPS matrix, providing protection against environmental stresses such as high salinity.
- Biomass: The total mass of living microorganisms (often measured as volatile suspended solids, VSS) in a treatment system responsible for pollutant degradation.
- Biomass washout: The unintended loss of active microbial biomass from a reactor, often due to high salinity or hydraulic shocks, leading to reduced treatment performance.
- COD: Chemical oxygen demand; a measure of organic pollutants in wastewater.
- Compatible solutes: Low-molecular-weight organic osmolytes (e.g., ectoine, glycine betaine) that counteract osmotic stress without inhibiting cytoplasmic enzymes.
- Conventional activated sludge: A suspended-growth biological treatment process relying on non-halophilic microbes, which performs poorly in high-salinity conditions.
- Conventional biological treatment systems: Standard aerobic or anaerobic processes designed for low-salinity wastewater, typically failing under high TDS due to osmotic stress on non-halophilic microbes.
- DAF: Dissolved air flotation; a primary physico-chemical treatment unit frequently placed upstream to remove oils, solids, and emulsified contaminants.
- Ectoine: A highly prevalent compatible solute synthesized or imported by moderate halophiles that helps cells maintain cytoplasmic water activity and adjust easily when salinity changes.
- EPS: Extracellular polymeric substances; polysaccharide and protein matrix produced by biofilms that shields cells from salinity and toxins.
- Extremophile: An organism, such as a halophile, adapted to thrive in extreme environmental conditions like high salinity.
- Halobacterium: An extreme halophilic archaeon that uses the salt-in strategy and is often studied for its adaptations to hypersaline environments.
- Halomonas: A genus of moderately halophilic bacteria widely used in saline wastewater treatment due to its robust salt tolerance and degradation capabilities.
- Haloferax: A genus of halophilic archaea known for its ability to degrade hydrocarbons and aromatics in high-salinity conditions.
- Halophilic: Salt-loving microorganisms requiring or tolerating high NaCl concentrations for growth.
- Halophilic IFAS/Biofilm System: Hybrid or attached bioreactor configurations that leverage halophilic microorganisms for effective treatment of high-salinity wastewater.
- HRT: Hydraulic retention time; the average time wastewater remains in the reactor, critical for achieving desired degradation.
- Hypersaline: Environments or wastewaters with extremely high salt concentrations, typically exceeding 5 to 15 % NaCl.
- IFAS: Integrated fixed-film activated sludge; hybrid reactor combining suspended flocs and attached biofilm on carriers.
- MBBR: Moving bed biofilm reactor; a biological treatment system utilizing high-surface-area carrier media to support robust halophilic biofilm growth and offer excellent biomass retention.
- Nitrification-denitrification: Biological nitrogen removal processes that are often challenging in saline conditions but can be supported in halophilic biofilm systems.
- Non-halophilic microbes: Microorganisms that lack adaptations to high salinity and typically experience plasmolysis or metabolic inhibition in saline wastewater.
- Ortho-cleavage: Ring-opening pathway of catechols by 1,2-dioxygenase, yielding muconic acid derivatives that feed the TCA cycle.
- Osmotic stress: The physiological strain exerted on microbes by high ionic strength environments, which can lead to cell dehydration, plasmolysis, and reduced metabolic activity in non-adapted species.
- Phenol: A common recalcitrant aromatic pollutant in industrial saline wastewater, serving as a model substrate for studying halophilic degradation pathways.
- Plasmolysis: Cell shrinkage due to water efflux under hyperosmotic conditions, a common issue for non-halophilic microbes in saline environments.
- Proteobacteria: A major bacterial phylum prevalent in saline treatment systems, including many halophilic degraders.
- Recalcitrant: Organic compounds that are difficult to degrade biologically due to their chemical structure, such as certain aromatics in saline wastewater.
- Salt-in strategy: Intracellular accumulation of KCl for osmotic balance, typical of extreme halophilic archaea.
- Salt-out strategy: A salt tolerance approach used predominantly by moderate halophiles and halotolerant bacteria, relying on the synthesis or import of compatible solutes rather than intracellular salt accumulation.
- Sequencing batch reactor: A fill-and-draw reactor configuration that allows flexible operation and is often used in hybrid halophilic treatment setups.
- Shock load: A sudden increase in salinity, organic load, or flow rate that can stress microbial communities in wastewater treatment systems.
- TDS: Total dissolved solids, primarily measured as NaCl equivalents in wastewater.
- Zwitterionic: Molecules carrying both positive and negative charges (example, many compatible solutes) that remain neutral overall and stabilize proteins in high-salt conditions.
Frequently Asked Questions
Q1: What is the energy cost of synthesizing compatible solutes, and how does this affect organic degradation rates compared to the "salt-in" strategy?
A: Synthesizing compatible solutes (like ectoine (C6H10N2O2)) is metabolically expensive, requiring about 20 to 30 ATP equivalents per molecule. However, paying this energy "tax" gives cells the flexibility to rapidly adjust their internal chemistry when salinity changes. In contrast, the "salt-in" strategy relies on ion pumps; this has a lower biosynthetic demand but restricts the microbes to stable, high-salt conditions. Despite the high energy cost of compatible solutes, strains like Halomonas thrive, often achieving steady phenol degradation rates of 0.5 to 1.0 g/g VSS·d at 10 % NaCl even during salinity fluctuations.
Q2: How do biofilms affect the degradation kinetics (like μ_max and K_s) of halophilic bacteria compared to suspended growth?
A: For a strain like Halomonas degrading phenol, the maximum specific growth rate (μ_max) is typically 0.15 to 0.25 h⁻¹, with a half-saturation constant (K_s) around 5 to 20 mg/L under ideal suspended conditions. In an IFAS biofilm, pollutants must physically penetrate the microbial matrix, causing a "diffusion limitation". This makes the apparent (K_s) higher by 20 to 50% because the inner bacteria effectively "see" less pollutant. However, the overall treatment rate still vastly improves because biofilms hold much more active biomass (typically 8 to 15 g VSS/L on carriers). To balance this diffusion limit, we use tools like the Thiele modulus to check mass transfer and choose an appropriate carrier media, often keeping biofilm thickness under 500 μm.
Q3: Why do halophilic IFAS systems exhibit superior nitrification resilience compared to conventional systems at 50 g/L TDS, and what microbial interactions underpin this?
A: Nitrifying bacteria are highly sensitive to salt, but biofilms offer them a unique physical shield. While they require oxygen and live in the aerated layers of the biofilm, the thick extracellular polymeric substance (EPS) matrix physically buffers them against sudden spikes in salinity. The EPS binds cations and slows down the diffusion of salt into the cells. Additionally, heterotrophic bacteria in the biofilm consume oxygen, creating low-oxygen zones deeper inside that allow for simultaneous denitrification. This structural teamwork allows halophilic systems to achieve > 80 % ammonia removal, whereas conventional suspended sludge often drops below 20 % at the same salinity.
Q4: What is the theoretical minimum hydraulic retention time (HRT) for phenol mineralization in a halophilic biofilm reactor, and how is it calculated?
A: We calculate this using the Monod rate equation combined with a steady-state mass balance. To find the rate of pollutant removal (r), we use the maximum growth rate (μ_max), the substrate concentration (S), active biomass (X), the half-saturation constant (K_s), and the biomass yield (Y):
Once we have the rate, the steady-state HRT balance is:
Let's plug in typical pilot values: μ_max= 0.2 h⁻¹, K_s= 10 mg/L, active biomass X= 15 g/L, inlet phenol S_in= 500 mg/L, and a target effluent Sout of 1 mg/L. Factoring in a typical yield (Y), the minimum HRT calculates to roughly 4 to 6 hours. Adding biomass decay rates gives an even more precise design equation, but this range closely matches real-world pilot study results.
Q5: How do genetic engineering approaches, such as the overexpression of ectoine genes, enhance stability in plants facing variable salinity?
A: Overexpressing the ectABC genes, which are the specific genes responsible for synthesizing the compatible solute ectoine, under salt-inducible promoters can raise intracellular ectoine levels by 2 to 3 times. This genetic boost essentially supercharges the microbes' "salt-out" strategy. As a result, it shortens the microbial recovery time after sudden salinity shocks by about 50 % and helps maintain over 90 % COD removal even when salt concentrations fluctuate wildly between 3 and 20 %. Combined with good process monitoring, these genetic enhancements support highly stable microbial communities over long operation periods.
Bibliography
Tackling high-salinity wastewater?
Contact Ecologix Environmental Systems today for engineered halophilic treatment solutions.
Contact Us

